A non-heme ferric dioxygenase catalyzing the intradiol cleavage of all the examined catechol derivatives, 3,5-dichlorocatechol, was reported in Pseudomonas cepacia CSV90, grown with 2,4-D as the sole carbon source. is reported to produce a high molecular weight C12O (100 kDa) that is stable at pH 8 but optimally active at pH 6.2. Pseudomonas aeruginosa TKU002 capable of mineralizing benzoic acid was reported to produce a low molecular weight C12O showing the highest activity against pyrogallol, which is an unusual characteristic. found to be dependent on Fe 2+ and Fe 3+ ions with high substrate specificity, showing molecular weight ranges from 22 to 35 kDa. Ĭ12O was first isolated and purified in Pseudomonas spp. C12O contains Iron(III) oxide as a prosthetic group, and it is part of the enzymes that cleave catechol via the ortho-cleavage, resulting in the formation of cis-cis-muconic acid. C12O is mostly reported in Gram-negative bacteria, but much less information is available about these enzymes in Gram-positive bacteria. The enzyme incorporates an oxygen atom into the catechol, resulting in the formation of cis-cis-muconic acid. Ĭatechol 1,2-dioxygenases have the potential to be used in the process of remediating wastewater contaminated with phenol, benzoate, fluorocatechol, bromocatechol, chlorocatechol, methylcatechol, herbicides (diuron), polychlorinated biphenyls, and chloroethanes. The final intermediates of both pathways then enter the tricarboxylic acid cycle. The catechol is then oxidized via an ortho-cleavage pathway by catechol 1,2-dioxygenase (C12O), or via a meta-pathway to 2-hydroxymuconic semialdehyde by catechol 2,3-dioxygenase (C23O) to open the ring. During the aerobic biodegradation of aromatic compounds, phenol, and it derivatives (e.g., 2,4-dichlorophenoxacetic acid (2,4-D) and 2,4-dichlorophenol (2,4-DCP)), catechols are formed as the central intermediates by the introduction of hydroxyl groups facilitated at ortho- or meta-positions. 
Microorganisms have developed mechanisms to degrade these compounds with the aid of enzymes. The widespread distribution of aromatic compounds in the environment has led to an increase in pollution, which affects the health quality of living organisms. The characterization and optimization of C12O activity can assist in understanding the 2,4-DCP metabolic pathway in PcUFB2 and its possible application in bioremediation strategies. ES-MS spectra of tryptic digested SDS-PAGE band and bioinformatics studies revealed that C12O shared 81% homology with homogentisate 1,2-dioxygenase reported in other Pseudomonas chlororaphis strains. The Lineweaver–Burk plot showed the v max and K m values of 16.67 µM/min and 35.76 µM, respectively. The enzyme was optimally active at pH 7.5 and a temperature of 30 ☌. A 35 kDa protein was purified to 1.5-fold with total yield of 13.02% by applying anion exchange and gel filtration chromatography. The crude cell extract showed 10.34 U/mL of C12O activity with a specific activity of 2.23 U/mg of protein. Preliminary studies showed that PcUFB2 could degrade 40 ppm of 2,4-dichlorophenol (2,4-DCP). The aim of this study was to purify, characterize, and predict the template-based three-dimensional structure of catechol 1,2-dioxygenase (C12O) from indigenous Pseudomonas chlororaphis strain UFB2 ( PcUFB2). 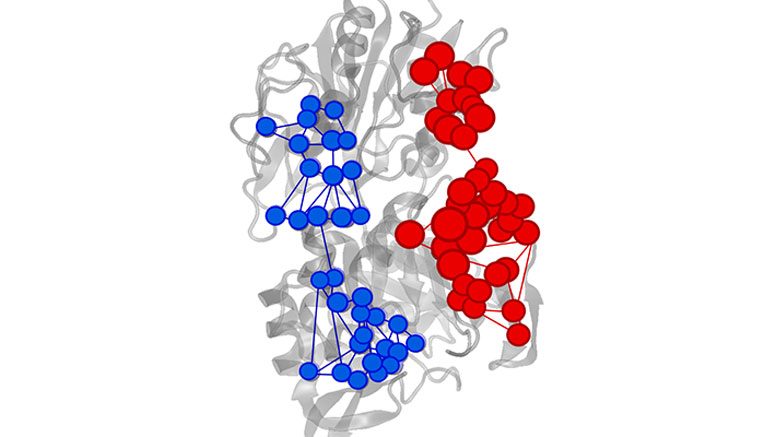
Catechol dioxygenases in microorganisms cleave catechol into cis- cis-muconic acid or 2-hydroxymuconic semialdehyde via the ortho- or meta-pathways, respectively. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
